NEW HAVEN, Conn., U.S.: We are learning more about SARS-CoV-2 every day, including more about how to effectively test for it. Researchers at the Yale School of Public Health have found that employing saliva samples to test for the virus could lower the risk of infection for health workers. The researchers have partnered with the National Basketball Association (NBA) to test the efficacy of a saliva sampling method that they say presents as a faster, less invasive and more accurate alternative to the recommended nasopharyngeal and oropharyngeal sampling methods.
The 2019–2020 NBA season is slated to resume in the city of Orlando in late July, but the results of a study involving players, coaches and staff from the league may have a greater impact on the American public than the results of the on-court fixtures.
Led by Drs. Nathan Grubaugh and Anne Wyllie, the research team will test select members of NBA teams and their staff and management for the virus using SalivaDirect. According to a Yale University press release, SalivaDirect reduces testing times by over 1 hour, costs less, exposes health care professionals to reduced risk, and could be effectively tested in a sporting environment.
“Because the NBA regularly tests players and on-court staff, who necessarily work in close contact without face coverings, the group of staff and players provides a unique opportunity for a comparison study to analyze the new test’s accuracy and sensitivity, which will enable the submission to the FDA [U.S. Food and Drug Administration] for SalivaDirect,” the researchers explained.
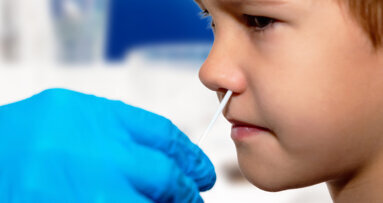
The sampling method is the result of an April study by the Yale school, which found that saliva samples taken from just inside the mouth provided both greater detection sensitivity and consistency throughout the various stages of COVID-19 and less variability in testing results, when compared with the recommended nasopharyngeal method. In the study, which had not been peer reviewed at the time of writing, SARS-CoV-2 was detected in two asymptomatic health care workers who had previously tested negative for the virus in a nasopharyngeal swab test. This led the researchers to hypothesize that saliva testing could be more effective in detecting mild infections.
“[Saliva] is sensitive for SARS-CoV-2 detection in both COVID-19 inpatients and asymptomatic health care worker screening”
– Dr Anne Wyllie, research scientist, Yale School of Public Health
Wyllie, the first author of the initial study and an associate research scientist in the epidemiology of microbial diseases at the Yale School of Public Health, told Dental Tribune International that the results had led to further investigation. “With the ongoing pandemic, this has been an ongoing study. We have recently updated our dataset and found the same trends—saliva is sensitive for SARS-CoV-2 detection in both COVID-19 inpatients and asymptomatic health care worker screening.”
She said that the response to the study had been overwhelming. “We have been advising institutions all over the country on how to work with saliva and validate it as a sample type. Being a noninvasive sample and able to be reliably self-collected, it appears key to meeting mass testing demands.”
The results of the NBA study are expected by the end of July and will be shared publicly.
Saliva sampling proposed for antibody testing for SARS-CoV-2
The authors of a May letter published in the British Dental Journal wrote that the use of saliva samples presents advantages in clinical settings. “It is less invasive and more convenient to patients as compared to [nasopharyngeal and/or oropharyngeal swabs] or blood samples (especially desirable in multiple testing for disease monitoring). Secondly, with clear instructions, patients can collect saliva themselves, thereby minimizing the risk of virus transmission to health care personnel and avoiding use of personal protective equipment.”
The authors pointed out that serological tests that detect IgM and IgG antibodies are a robust method for detecting current and past SARS-CoV-2 infections and that preliminary studies had demonstrated the production of SARS-CoV-specific secretory IgA in the saliva of animals. “Hence, it is reasonable to speculate that anti-COVID-19 antibodies might also be present in human saliva although this has not been reported nor has their potential use for diagnostics and disease monitoring. This clearly warrants future studies,” they concluded.
In early May, the FDA gave authorization for the first diagnostic test that uses saliva samples to test for SARS-CoV-2. The test is made by the Clinical Genomics laboratory of Rutgers New Jersey Medical School, and the saliva sample can be collected by patients in their homes using the Spectrum Solutions SDNA-1000 saliva collection device.
Tags:



 Austria / Österreich
Austria / Österreich
 Bosnia and Herzegovina / Босна и Херцеговина
Bosnia and Herzegovina / Босна и Херцеговина
 Bulgaria / България
Bulgaria / България
 Croatia / Hrvatska
Croatia / Hrvatska
 Czech Republic & Slovakia / Česká republika & Slovensko
Czech Republic & Slovakia / Česká republika & Slovensko
 France / France
France / France
 Germany / Deutschland
Germany / Deutschland
 Greece / ΕΛΛΑΔΑ
Greece / ΕΛΛΑΔΑ
 Hungary / Hungary
Hungary / Hungary
 Italy / Italia
Italy / Italia
 Netherlands / Nederland
Netherlands / Nederland
 Nordic / Nordic
Nordic / Nordic
 Poland / Polska
Poland / Polska
 Portugal / Portugal
Portugal / Portugal
 Romania & Moldova / România & Moldova
Romania & Moldova / România & Moldova
 Slovenia / Slovenija
Slovenia / Slovenija
 Serbia & Montenegro / Србија и Црна Гора
Serbia & Montenegro / Србија и Црна Гора
 Spain / España
Spain / España
 Switzerland / Schweiz
Switzerland / Schweiz
 Turkey / Türkiye
Turkey / Türkiye
 UK & Ireland / UK & Ireland
UK & Ireland / UK & Ireland
 International / International
International / International
 Brazil / Brasil
Brazil / Brasil
 Canada / Canada
Canada / Canada
 Latin America / Latinoamérica
Latin America / Latinoamérica
 USA / USA
USA / USA
 China / 中国
China / 中国
 India / भारत गणराज्य
India / भारत गणराज्य
 Pakistan / Pākistān
Pakistan / Pākistān
 Vietnam / Việt Nam
Vietnam / Việt Nam
 ASEAN / ASEAN
ASEAN / ASEAN
 Israel / מְדִינַת יִשְׂרָאֵל
Israel / מְדִינַת יִשְׂרָאֵל
 Algeria, Morocco & Tunisia / الجزائر والمغرب وتونس
Algeria, Morocco & Tunisia / الجزائر والمغرب وتونس
 Middle East / Middle East
Middle East / Middle East

























To post a reply please login or register